13 May Acids and Bases
Students and parents often complain about Chemistry being a tough nut to crack. They find the teaching methods by the various teachers in Singapore to be bland and lacklustre. Miracle Learning Centre offers help in A Level Chemistry, O Level Chemistry and JC Chemistry to inspire the students about the subject. With the best teachers from the tuition industry of Singapore we have made a team and a course which not only inspires the students but builds their self confidence by making the complex topics easy-peasy for them.
One of the most important and critical topic of Chemistry is Acids and Bases. They play a central role in Chemistry as other than redox reactions, every chemical reaction can be classified as an acid-base reaction. The term acid was first used in the seventeenth century which comes from the Latin root ac-, meaning “sharp”.
Thereare many definitions to acids and bases. The most useful is termed as Lewis Acids and Bases. According to the theory, acids react with bases to share electrons, without any change in the oxidation numbers of atoms.
Simple examples of acidic solutions are an orange juice or cola. On the other hand, having egg whites or even baking soda, gives the basic concept of bases. It may be noticed that acidic materials generally taste sour, or basic objects like soap or bleach are slippery. The reason to this is that an acidic solution has a high concentration of hydrogen ions, greater than that of pure water and a basic solution has a lower concentration of hydrogen ions, less than that of pure water.
With the above description, it is well understood that acidic and basic solutions are classified based on the concentration of hydrogen ions in relation to pure water. Acidic solutions have a high concentration H+ than water (>1*10-7 M), whereas alkaline solutions have a lower H+concentration (<1*10-7 M).
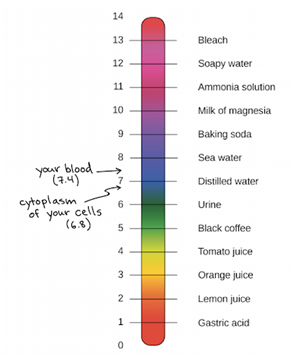
The pH Scale
pH is the amount of acidity or alkalinity in a solution. It is the measure of protons or hydrogen ions that are present in an aqueous solution. To rank solutions based on acidity or alkalinity the pH scale is used. The scale lies within the range of 0 to 14. Any solution below 7.0 is acidic, and solutions above 7.0 is alkaline or basic. The human stomach it is highly acidic with a pH level of 1 to 2. Stomach cells, mainly those that come in direct contact with stomach acid and food, are constantly get substituted by new ones. Studies show that the lining of human stomach is replaced every seven to ten days.